
8 tips to follow when choosing a restriction enzyme for in vitro mRNA vaccine production
Posted on Friday, September 24, 2021
By
Topic: What is Trending in Science
When it comes to generating mRNA vaccines, you should start how you want to finish – with optimal performance. The first step in in vitro transcription (IVT) relies on choosing the best fit restriction enzyme (RE) to linearize a template plasmid. As the leading scientific resource for enzyme characterization, quality, and manufacturing, New England Biolabs® shares practical tips on RE qualities for IVT.
The COVID-19 pandemic has necessitated the creation of vaccines at record speed, and mRNA-based vaccines have emerged as alternatives to conventional vaccine approaches. mRNA expression is transient and rapid. It is easily synthesized, standardized and scaled up - these are advantageous features because they streamline a lot of discovery and early development work. Once the viral genome is sequenced, the DNA sequence of interest (that could potentially be used as a vaccine target) can be identified within a few days. It’s been an extraordinary scientific success story.
The process of making an mRNA vaccine is enzymatic in nature: after a plasmid containing the vaccine target sequence is transformed into bacteria, fermented on a large scale and then harvested, the workflow utilizes several enzymes. The plasmid is linearized with a restriction enzyme, the DNA is then used as a template for in vitro transcription to produce mRNA. In addition, mRNA requires either cotranscriptional or enzymatic capping as well as a poly(A) tail to be functional. The resultant mRNA will ultimately be mixed with lipid nanoparticles and injected as a vaccine, thereby producing the antigen required to elicit an immune response. And, while the mRNA synthesis process has been around for a long time and used in many biotechnology applications, synthetic mRNA was only shown to elicit an immune response in 2012 and tested as a vaccine against the Zika virus in 2017. The requirements for enzymes used to manufacture mRNA in therapeutics and for generating vaccines are a little different.
A simplified workflow for mRNA vaccine production utilizes several enzymes, including restriction enzymes.
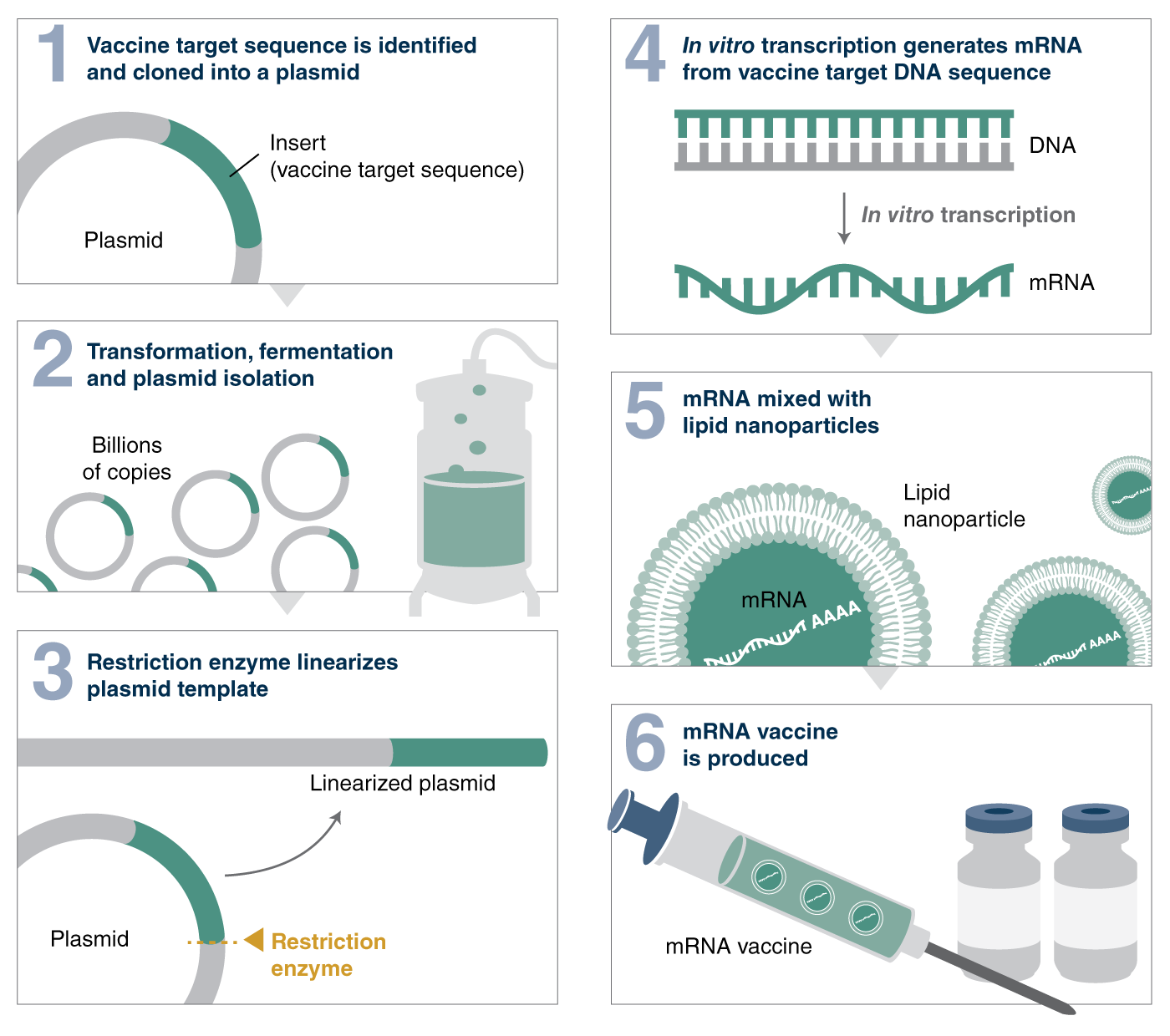
One of the first critical steps in making the SARS-CoV-2 vaccine, for example, is to linearize the plasmid DNA that contains the gene encoding the viral spike protein. Restriction enzymes are used to linearize the template plasmids. Therefore, it is important to ensure restriction enzyme manufacturing and formulation are performed in an animal-free environment and adhere to the highest quality standards. NEB has been involved in the research and manufacturing of restriction enzymes for almost 50 years – our expertise in this area allows us to provide the following guidance and things to consider when selecting a restriction enzyme for vaccine development:
(1) Animal-free manufacturing removes risks - Animal-derived materials, such as heparin, are often used in the purification of restriction enzymes. NEB offers restriction enzymes that are processed and/or formulated without animal-derived products.
(2) Animal-free formulation - Albumins, such as BSA, help stabilize enzymes when added to final formulations. NEB offers final formulations with recombinant Albumin (rAlbumin), ensuring no animal products are present to support FDA compliant manufacturing down the line.
(3) Longer recognition sequence for minimal plasmid processing - The longer the recognition sequence, the less likely the sequence will appear in the gene fragment, simplifying manufacturing of the plasmid template. It is therefore desirable to select restriction enzymes with longer recognition sequences. NEB offers many enzymes with 7-8 base recognition sequences.
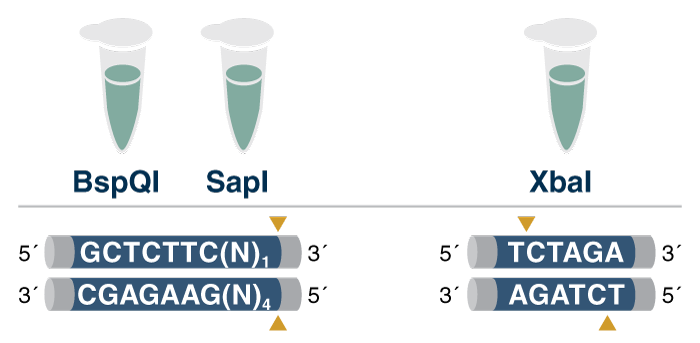
(4) Cleavage product - Additionally, the restriction enzyme should not leave a 3´ overhang. The presence of 3´ overhangs can lead to increased production of spurious by-products from the template during in vitro transcription (IVT).
(5) Stability of enzyme for optimal performance - Some enzymes are more stable than others. When selecting an enzyme, make sure you find the most stable enzyme possible (24-month expiration and -20°C storage preferred). Watch out for enzymes that have shorter shelf-life and -80°C storage recommendations*.
*In some instances, new NEB products may state a 12-month shelf-life before 24-month real-time stability is complete.
(6) Incubation temperature affects individual enzymes activities - Most restriction enzymes recommend incubation temperatures at 37°C. Be aware that some enzymes require alternate temperatures. This table lists the activity at 37°C should you require a specific enzyme for your construct.
(7) Detergents should comply with local regulations - Some enzymes contain small amounts of detergent, e.g., Triton-X-100, to stabilize the enzyme. It’s important to review the formulation components of your desired enzyme to ensure the levels of detergent do not conflict with local regulations, e.g., REACH in Europe.
(8) Type IIS Restriction Enzymes for a 'scarless' template - Type IIS restriction enzymes are the enzyme of choice for the linearization of plasmids used in vaccine development because they cut outside their recognition sequence and ensure the integrity of the poly(A) tail encoded by the manufacturer. Using a Type IIS RE, no “scar” remains on the DNA template after linearization, and no unwanted nucleotides are added during IVT.
- NEB offers over 30 Type IIS Restriction Enzymes, including BspQI, an isoschizomer of LguI, now available in GMP-grade*
- Multi-site enzymes. Some enzymes require more than one site for efficient cleavage. Often, the recommendation is to add an oligo to the reaction, which will require removal. Learn more here.
The following enzymes are popular candidates for customers considering template generation with a restriction enzyme:
GMP-grade BspQI formulation now available.
For more information on enzymes for your IVT process, including our GMP-grade IVT portfolio for therapeutic RNA manufacturing, please contact our Customized Solutions team. NEB has expert knowledge in finding the best restriction enzyme for your needs.
* "GMP-grade" is a branding term NEB uses to describe reagents manufactured at NEB’s Rowley facility. The Rowley facility was designed to manufacture reagents under more rigorous infrastructure and process controls to achieve more stringent product specifications and customer requirements. Reagents manufactured at NEB’s Rowley facility are manufactured in compliance with ISO 9001 and ISO 13485 quality management system standards. However, at this time, NEB does not manufacture or sell products known as Active Pharmaceutical Ingredients (APIs), nor does NEB manufacture its products in compliance with all of the Current Good Manufacturing Practice regulations.
NEB will not rent, sell or otherwise transfer your data to a third party for monetary consideration. See our Privacy Policy for details. View our Community Guidelines.
Products and content are covered by one or more patents, trademarks and/or copyrights owned or controlled by New England Biolabs, Inc (NEB). The use of trademark symbols does not necessarily indicate that the name is trademarked in the country where it is being read; it indicates where the content was originally developed. See www.neb.com/trademarks. The use of these products may require you to obtain additional third-party intellectual property rights for certain applications. For more information, please email busdev@neb.com.
Don’t miss out on our latest NEBinspired blog releases!
- Sign up to receive our e-newsletter
- Download your favorite feed reader and subscribe to our RSS feed
Be a part of NEBinspired! Submit your idea to have it featured in our blog.





